How COVID-19 vaccine technology may help skin cancer patients fight melanoma

Drug company Moderna recently announced that its experimental cancer vaccine – based on the messenger RNA (mRNA) technology used in some COVID-19 vaccines – has been shown to work against melanoma.
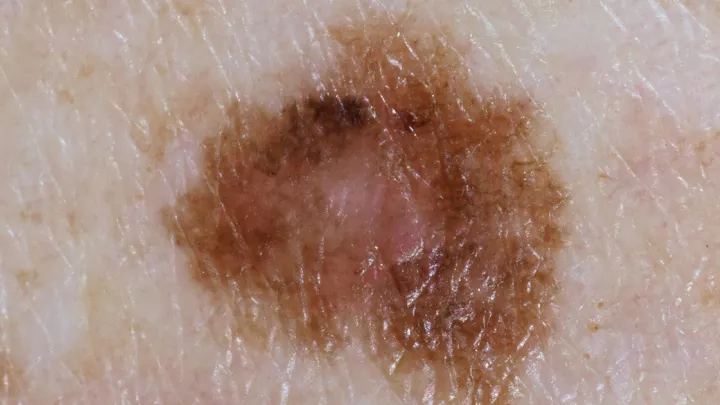
Cases of melanoma have been increasing steadily for the past 30 years, especially among teens and young adults. And while it accounts for just 1% of all skin cancers, melanoma is responsible for the majority of skin cancer-related deaths.
According to the announcement, when Moderna’s vaccine was combined with Keytruda®, an immunotherapy drug made by Merck, it reduced the risk of recurrence or death from melanoma by 44% compared with Keytruda alone in a recent trial.
How mRNA vaccines help fight cancer
“We are already using immunotherapies like Keytruda® to treat melanoma,” says surgical oncologist Joshua Mammen, MD, PhD. “And while they’re successful in revving up the immune system to fight cancer, we are realizing that combining them with another medication may make them even more effective.”
To build the vaccine, researchers took samples of patients' tumors and healthy tissue. They then analyzed the samples to decode their genetic sequence and isolate mutant proteins associated only with the cancer. Finally, they used that information to design a tailor-made cancer vaccine.
When the vaccine is injected, the patient's cells act as a manufacturing plant. It produces perfect copies of the mutations for the immune system to recognize and destroy.
Are mRNA cancer vaccines safe?
The mRNA COVID-19 vaccines were developed quickly because of the decades of work already done on cancer vaccines. In that period, immunotherapy emerged as a new approach to treating cancer. Immunotherapy has become an effective treatment for many patients.
The mRNA vaccines, such as those used for COVID-19, have proven to be exceedingly safe thus far. “We’re still waiting for a more formal presentation of the safety data from the cancer vaccine study,” says Dr. Mammen. “But, side effects are likely similar to current immunotherapy treatments. For example, current immunotherapy treatment can affect an individual’s thyroid and adrenal glands.”
When will the melanoma vaccine be available?
It’s too early to say when a vaccine will be ready to treat melanoma outside of a clinical trial. Moderna’s cancer vaccine just completed its phase 2 trial, which is the first step in testing its safety and effectiveness. Phase 3 clinical trials, expected to begin sometime in 2023, are where the vaccine will be tested on a larger scale.
“One thing they’ll be looking for in phase 3 clinical trials is whether the cancer returns,” explains Dr. Mammen. “Unfortunately, while immunotherapy treatments are very effective and have fewer side effects, the cancer can return after a year or two in some individuals. Our hope is that the addition of the cancer vaccine would help the body target and eliminate stray melanoma cells, making it even less likely for the cancer to come back.”
Can I participate in a clinical trial?
“Interested individuals may be able to participate in phase 3 trials of the cancer vaccine at Nebraska Medical Center,” says Dr. Mammen. “We already use immunotherapy treatments, including Keytruda®, to treat patients with melanoma. If we do participate in clinical trials, we’d essentially be adding the cancer (mRNA) vaccine to already existing treatments.”
“Advances in cancer treatment are made possible by generous individuals willing to participate in these studies,” adds Dr. Mammen. “For anyone interested, there are many clinical trials for melanoma and other cancers at the Fred & Pamela Buffett Cancer Center.”